Science Highlights Archives
New Technique Blasts Circulating Cancer Cells in the Blood
 Cancer cells can spread to new areas of the body, often through the blood, and causes nearly 90% of cancer-related deaths. Numerous lives would be saved if cancer cells that break away from a primary tumor could be detected and destroyed in the blood before the cells spread and form new tumors. Lihong Wang, a 2012 Pioneer and 2013 Transformative Researcher, has developed a method that promises to do just that. Using the unique optical characteristics of skin cancer-causing melanoma cells, Wang and his team developed lasers that can detect and target melanoma cells circulating in the blood and vaporize them without harming surrounding tissue. Preliminary studies are encouraging and provide hope that the technique may be useful in preventing the spread of cancer throughout the body.
Cancer cells can spread to new areas of the body, often through the blood, and causes nearly 90% of cancer-related deaths. Numerous lives would be saved if cancer cells that break away from a primary tumor could be detected and destroyed in the blood before the cells spread and form new tumors. Lihong Wang, a 2012 Pioneer and 2013 Transformative Researcher, has developed a method that promises to do just that. Using the unique optical characteristics of skin cancer-causing melanoma cells, Wang and his team developed lasers that can detect and target melanoma cells circulating in the blood and vaporize them without harming surrounding tissue. Preliminary studies are encouraging and provide hope that the technique may be useful in preventing the spread of cancer throughout the body.
Visualizing RNA using Expansion Microscopy
 RNA molecules act as messengers, carrying copies of genetic instructions from the DNA throughout the cell and are crucial for protein synthesis and proper cell functioning. Arjun Raj (a 2011 New Innovator) and Edward Boyden (a 2007 New Innovator, 2012 and 2013 Transformative Researcher, and 2013 Pioneer) describe a new technique that can image RNA in cells and tissue by using “expansion microscopy,” a method where samples are physically enlarged with an expandable polymer, making high resolution imaging possible with common laboratory microscopes. Knowing the distribution of RNA inside cells will enable scientists to better understand how cells control gene expression and could aid study of diseases caused by the improper movement of RNA.
RNA molecules act as messengers, carrying copies of genetic instructions from the DNA throughout the cell and are crucial for protein synthesis and proper cell functioning. Arjun Raj (a 2011 New Innovator) and Edward Boyden (a 2007 New Innovator, 2012 and 2013 Transformative Researcher, and 2013 Pioneer) describe a new technique that can image RNA in cells and tissue by using “expansion microscopy,” a method where samples are physically enlarged with an expandable polymer, making high resolution imaging possible with common laboratory microscopes. Knowing the distribution of RNA inside cells will enable scientists to better understand how cells control gene expression and could aid study of diseases caused by the improper movement of RNA.
Ultra-Resolution Microscopy
 Peng Yin, a 2010 New Innovator and 2013 Transformative Research awardee, enhanced DNA nanotechnology-powered super-resolution microscopy to visualize distinct molecules only five nanometers apart. The ultra-high resolution is achieved through a novel method that compensates for movements of the microscope stage and by further harnessing the power of DNA-PAINT, which is based on the transient binding of short strands of complementary DNA, one which is tagged with a fluorescent dye and the other is attached to the molecule of interest. The method opens the way for researchers to study individual molecular features in molecular complexes that may ultimately lead to a greater understanding of their functions.
Peng Yin, a 2010 New Innovator and 2013 Transformative Research awardee, enhanced DNA nanotechnology-powered super-resolution microscopy to visualize distinct molecules only five nanometers apart. The ultra-high resolution is achieved through a novel method that compensates for movements of the microscope stage and by further harnessing the power of DNA-PAINT, which is based on the transient binding of short strands of complementary DNA, one which is tagged with a fluorescent dye and the other is attached to the molecule of interest. The method opens the way for researchers to study individual molecular features in molecular complexes that may ultimately lead to a greater understanding of their functions.
Treating Mitochondrial Disease with Hypoxia
 Vamsi Mootha (2011 Transformative Researcher) and Feng Zhang (2010 and 2015 Transformative Researcher and 2012 Pioneer) published a paper on using low levels of oxygen to treat mitochondrial diseases, which are debilitating and largely untreatable. The mitochondrial respiratory chain uses oxygen to generate cellular energy, and impairments to it can cause blindness, deafness, and disease in the brain, heart, muscles, stomach, blood, liver, and kidney. However, endogenous mechanisms may exist to help cells cope with mitochondrial defects. Mootha et al. identified the hypoxia response, a mechanism that helps cells adapt to low oxygen levels, as an effective treatment to mitochondrial dysfunction. Zebrafish and mouse models of the mitochondrial disease Leigh syndrome showed fewer symptoms and had a dramatically longer life span when raised in a low oxygen environment. Chronic hypoxia increased survival, body weight, body temperature, behavior, neuropathology, and disease in the mouse model. While further studies are needed to assess the safety of using hypoxic conditions to treat human diseases associated with mitochondrial dysfunction, it may be that the body’s naturally evolved response to low oxygen may be protective against mitochondrial disease.
Vamsi Mootha (2011 Transformative Researcher) and Feng Zhang (2010 and 2015 Transformative Researcher and 2012 Pioneer) published a paper on using low levels of oxygen to treat mitochondrial diseases, which are debilitating and largely untreatable. The mitochondrial respiratory chain uses oxygen to generate cellular energy, and impairments to it can cause blindness, deafness, and disease in the brain, heart, muscles, stomach, blood, liver, and kidney. However, endogenous mechanisms may exist to help cells cope with mitochondrial defects. Mootha et al. identified the hypoxia response, a mechanism that helps cells adapt to low oxygen levels, as an effective treatment to mitochondrial dysfunction. Zebrafish and mouse models of the mitochondrial disease Leigh syndrome showed fewer symptoms and had a dramatically longer life span when raised in a low oxygen environment. Chronic hypoxia increased survival, body weight, body temperature, behavior, neuropathology, and disease in the mouse model. While further studies are needed to assess the safety of using hypoxic conditions to treat human diseases associated with mitochondrial dysfunction, it may be that the body’s naturally evolved response to low oxygen may be protective against mitochondrial disease.
Manipulating the Electron Transport Chain
 Vamsi Mootha, a 2011 Transformative Researcher, created a nicotinamide adenine dinucleotide (NAD+) producing bacteria Lactobacillus brevis (LbNOX) for manipulating the crucial electron transport system. Disruptions in electron transport chain activity in mitochondria are associated with many diseases, and changes in the ratio of NAD+ and its reduced form NADH play a crucial role in key metabolic fluxes, gluconeogenesis, and signaling on the cytosolic or mitochondrial NAD+/NADH ratios. Mootha et al. found increasing NAD+ in the cytosol or mitochondria improved proliferative and metabolic defects caused by an impaired electron transport chain and demonstrates the utility of LbNOX for manipulating redox states in specific targets.
Vamsi Mootha, a 2011 Transformative Researcher, created a nicotinamide adenine dinucleotide (NAD+) producing bacteria Lactobacillus brevis (LbNOX) for manipulating the crucial electron transport system. Disruptions in electron transport chain activity in mitochondria are associated with many diseases, and changes in the ratio of NAD+ and its reduced form NADH play a crucial role in key metabolic fluxes, gluconeogenesis, and signaling on the cytosolic or mitochondrial NAD+/NADH ratios. Mootha et al. found increasing NAD+ in the cytosol or mitochondria improved proliferative and metabolic defects caused by an impaired electron transport chain and demonstrates the utility of LbNOX for manipulating redox states in specific targets.
Counting Molecules with qPAINT
 Peng Yin (2010 New Innovator and 2013 Transformative Researcher) and Avital Rodal (2012 New Innovator) published a paper in Nature Methods describing enhancements in optical super-resolution microscopy for counting molecules in complexes. Quantitative points accumulation in nanoscale topography (qPAINT) improves DNA-PAINT by working independently from dye photophysics for robust counting with high precision and accuracy over a wide dynamic range. The method is immune to photobleaching and is capable of multiplexing for a large number of targets.
Peng Yin (2010 New Innovator and 2013 Transformative Researcher) and Avital Rodal (2012 New Innovator) published a paper in Nature Methods describing enhancements in optical super-resolution microscopy for counting molecules in complexes. Quantitative points accumulation in nanoscale topography (qPAINT) improves DNA-PAINT by working independently from dye photophysics for robust counting with high precision and accuracy over a wide dynamic range. The method is immune to photobleaching and is capable of multiplexing for a large number of targets.
Using Gene Editing to Treat Duchenne Muscular Dystrophy
 Amy Wagers (2008 New Innovator), George Church (2013 Transformative Researcher), and Feng Zhang (2012 Pioneer; 2010 & 2015 Transformative Researcher) published back-to-back Science papers on using the CRISPR-Cas9 system to edit the genome of a mouse model of Duchenne muscular dystrophy and improving muscle function. The researchers loaded a virus with the DNA-cutting tool CRISPR and infected the mice's muscle cells. CRISPR excised a defective stretch of DNA, shortening the dystrophin protein and making it functional. The functional protein led to muscle improvement and increased strength in the mice and brings hope for one day using gene editing to treat human's with Duchenne Muscular Dystrophy.
Amy Wagers (2008 New Innovator), George Church (2013 Transformative Researcher), and Feng Zhang (2012 Pioneer; 2010 & 2015 Transformative Researcher) published back-to-back Science papers on using the CRISPR-Cas9 system to edit the genome of a mouse model of Duchenne muscular dystrophy and improving muscle function. The researchers loaded a virus with the DNA-cutting tool CRISPR and infected the mice's muscle cells. CRISPR excised a defective stretch of DNA, shortening the dystrophin protein and making it functional. The functional protein led to muscle improvement and increased strength in the mice and brings hope for one day using gene editing to treat human's with Duchenne Muscular Dystrophy.
The Need to Study Pathogen Growth In Vivo
 Dianne Newman, a 2012 Transformative Researcher, published a paper measuring the in vivo growth rate of Staphylococcus aureus in the sputum of pediatric patients with cystic fibrosis. Using a combination of isotope ratio mass spectrometry and heavy water labeling, Newman found S. aureus has a generation time of 2.1 days (two orders of magnitude slower than when grown in the laboratory) and is extremely heterogeneous at the single-cell level. The findings underscore the need to study slow growth physiology in pathogens to gain insight into their survival mechanisms, which will lead to improved drug design and clinical outcomes.
Dianne Newman, a 2012 Transformative Researcher, published a paper measuring the in vivo growth rate of Staphylococcus aureus in the sputum of pediatric patients with cystic fibrosis. Using a combination of isotope ratio mass spectrometry and heavy water labeling, Newman found S. aureus has a generation time of 2.1 days (two orders of magnitude slower than when grown in the laboratory) and is extremely heterogeneous at the single-cell level. The findings underscore the need to study slow growth physiology in pathogens to gain insight into their survival mechanisms, which will lead to improved drug design and clinical outcomes.
Wake-Up Call to Drug Resistant Tuberculosis
 Sanjay Jain, a 2009 New Innovator and 2014 Transformative Researcher, reported successfully treating a child with extensively drug-resistant TB, or XDR-TB, in the United States. Treatment was difficult, even with vast resources available. Jain was able to successfully monitor and treat the young patient but warns the case is "a wake-up call to the realities of TB."
Sanjay Jain, a 2009 New Innovator and 2014 Transformative Researcher, reported successfully treating a child with extensively drug-resistant TB, or XDR-TB, in the United States. Treatment was difficult, even with vast resources available. Jain was able to successfully monitor and treat the young patient but warns the case is "a wake-up call to the realities of TB."
mRNA Structure Rearrangement and Ribosome Movement
 Joseph Puglisi, a 2011 Transformative Research awardee, determined why ribosomes translating a particular bacteriophage T4 mRNA bypass a region of 50 nucleotides and resume translating the 3’ of the gap. Using single-molecule fluorescence with zero-mode waveguides to track individual Escherichia coli ribosomes during translation, Puglisi provides a mechanistic and conformational framework for bypassing, highlighting a non-canonical ribosomal state to allow for mRNA structure refolding to drive large-scale ribosome movements.
Joseph Puglisi, a 2011 Transformative Research awardee, determined why ribosomes translating a particular bacteriophage T4 mRNA bypass a region of 50 nucleotides and resume translating the 3’ of the gap. Using single-molecule fluorescence with zero-mode waveguides to track individual Escherichia coli ribosomes during translation, Puglisi provides a mechanistic and conformational framework for bypassing, highlighting a non-canonical ribosomal state to allow for mRNA structure refolding to drive large-scale ribosome movements.
Anesthesia's Effects on the Brain Differ in the Elderly and in Children
 Patrick Purdon (2009 New Innovator) and Emery Brown (2012 Transformative Researcher and 2007 Pioneer) released four papers on brain activity under anesthesia in children and the elderly that address how to improve anesthesia care and advance the neuroscience of development and aging. They found anesthesia-induced brain dynamics mirrors brain development in children, where different brain wave patterns under anesthesia appear to “turn on” at particular ages, coinciding with known brain developmental milestones. A similar effect is seen in the elderly but in reverse—certain brain waves seem to fade away in a manner consistent with known patterns of brain aging. Elderly patients are much more sensitive to anesthetic drugs than current age-adjusted dosing guidelines suggest, and the elderly enter a coma-like state (associated with poor cognitive outcomes) at much lower doses than with young patients. Elderly animals emerge from anesthesia much more slowly than younger animals, but the drug methylphenidate accelerates emergence. Purdon and Brown also found commercially available EEG-based anesthetic monitors (developed for adults) seem inaccurate when applied to children and the elderly and suggest new age-specific monitoring guidelines.
Patrick Purdon (2009 New Innovator) and Emery Brown (2012 Transformative Researcher and 2007 Pioneer) released four papers on brain activity under anesthesia in children and the elderly that address how to improve anesthesia care and advance the neuroscience of development and aging. They found anesthesia-induced brain dynamics mirrors brain development in children, where different brain wave patterns under anesthesia appear to “turn on” at particular ages, coinciding with known brain developmental milestones. A similar effect is seen in the elderly but in reverse—certain brain waves seem to fade away in a manner consistent with known patterns of brain aging. Elderly patients are much more sensitive to anesthetic drugs than current age-adjusted dosing guidelines suggest, and the elderly enter a coma-like state (associated with poor cognitive outcomes) at much lower doses than with young patients. Elderly animals emerge from anesthesia much more slowly than younger animals, but the drug methylphenidate accelerates emergence. Purdon and Brown also found commercially available EEG-based anesthetic monitors (developed for adults) seem inaccurate when applied to children and the elderly and suggest new age-specific monitoring guidelines.
Expanding Telomeres
 Helen Blau, a 2012 awardee, developed a new technique for rapidly extending telomeres and increasing the capacity of cells to divide. The researchers used transient delivery of non-immunogenic modified mRNA encoding the telomerase protein TERT to extend telomeres. This technology holds potential for investigating telomere extension to increase lifespan, as well as increasing cell numbers for research, including testing new drugs. Unlike previous methods of extending telomeres, it is temporary and does not cause cells to become immortalized. This technique also meets other criteria for safe and useful telomere extension therapy--it is non-immunogenic, has no risk of genomic insertion, and is rapid.
Helen Blau, a 2012 awardee, developed a new technique for rapidly extending telomeres and increasing the capacity of cells to divide. The researchers used transient delivery of non-immunogenic modified mRNA encoding the telomerase protein TERT to extend telomeres. This technology holds potential for investigating telomere extension to increase lifespan, as well as increasing cell numbers for research, including testing new drugs. Unlike previous methods of extending telomeres, it is temporary and does not cause cells to become immortalized. This technique also meets other criteria for safe and useful telomere extension therapy--it is non-immunogenic, has no risk of genomic insertion, and is rapid.
Reference:
Transient delivery of modified mRNA encoding TERT rapidly extends telomeres in human cells. Ramunas J, et al. FASEB J. 2015 May;29(5):1930-9.
Breakthrough Technique Expands the Capabilities of Microscopes
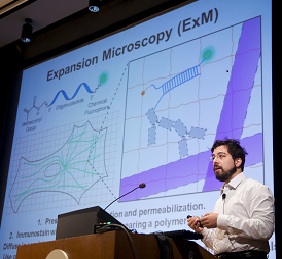 Microscopes allow researchers to peer into tiny cells and tissues by making the objects appear larger. However, visualizing extremely tiny cellular structures requires specialized microscopes that are very expensive, and even the most advanced microscopes are limited in how much they can magnify objects. Using a paradigm-shifting technique called expansion microscopy (ExM), Pioneer and Transformative Research Awardee Dr. Edward Boyden is taking the novel approach of actually making the samples themselves larger. Using a material commonly found in baby diapers, Dr. Boyden and colleagues at the Massachusetts Institute of Technology (MIT) are able to expand tissues to about 4.5 times their normal size, enabling visualization of tiny cellular structures and proteins with ordinary microscopes. This material, called sodium acrylate, holds proteins in place while it swells in the presence of water, resulting in minimal distortion of the sample’s structure. Using ExM, researchers could image a 3-dimensional slice of brain tissue and could zoom in to see the tiny synapse where neurons communicate with each other. By eliminating the need for costly, specialized equipment, ExM has the potential to enable many more scientists to examine structures otherwise unobservable in the cell.
Microscopes allow researchers to peer into tiny cells and tissues by making the objects appear larger. However, visualizing extremely tiny cellular structures requires specialized microscopes that are very expensive, and even the most advanced microscopes are limited in how much they can magnify objects. Using a paradigm-shifting technique called expansion microscopy (ExM), Pioneer and Transformative Research Awardee Dr. Edward Boyden is taking the novel approach of actually making the samples themselves larger. Using a material commonly found in baby diapers, Dr. Boyden and colleagues at the Massachusetts Institute of Technology (MIT) are able to expand tissues to about 4.5 times their normal size, enabling visualization of tiny cellular structures and proteins with ordinary microscopes. This material, called sodium acrylate, holds proteins in place while it swells in the presence of water, resulting in minimal distortion of the sample’s structure. Using ExM, researchers could image a 3-dimensional slice of brain tissue and could zoom in to see the tiny synapse where neurons communicate with each other. By eliminating the need for costly, specialized equipment, ExM has the potential to enable many more scientists to examine structures otherwise unobservable in the cell.
(Photo: Dr. Edward Boyden presenting ExM at the Common Fund's High Risk, High Reward Research Symposium in December 2014)
Listen to Dr. Boyden’s talk at the Common Fund’s High Risk, High Reward Research Symposium (starts at 1 hour, 33 minutes)
Read the MIT press release
Reference:
Chen F, Tillberg PW, and Boyden ES. Expansion Microscopy. Science, Jan 2014, DOI: 10.1126/science.1260088.
Transformative Research Awardee Uses New Technology to Discover Antibiotics
 Using a newly developed technology, a research team led by Dr. Kim Lewis of Northeastern University in Boston has discovered 25 new antibiotics, with one in particular very promising for potential future use to treat an array of difficult to treat human infections including MRSA (Methicillin-resistant Staphylococcus aureus), which in 2013 alone caused 480,000 infections. The Transformative Research Award was created specifically to support exceptionally innovative and/or unconventional research projects that have the potential to create or overturn fundamental paradigms. Although inherently risky, projects also have high potential to create breakthroughs that have the ability to change the landscape of a scientific area. (Photo: Slava Epstein/Northeastern University)
Using a newly developed technology, a research team led by Dr. Kim Lewis of Northeastern University in Boston has discovered 25 new antibiotics, with one in particular very promising for potential future use to treat an array of difficult to treat human infections including MRSA (Methicillin-resistant Staphylococcus aureus), which in 2013 alone caused 480,000 infections. The Transformative Research Award was created specifically to support exceptionally innovative and/or unconventional research projects that have the potential to create or overturn fundamental paradigms. Although inherently risky, projects also have high potential to create breakthroughs that have the ability to change the landscape of a scientific area. (Photo: Slava Epstein/Northeastern University)
For more information visit:
Two HRHR researchers awarded world’s largest brain research prize

 Two NIH Common Fund High Risk-High Reward Awardees were named as winners of The Brain Prize for 2013, which is awarded by the Grete Lundbeck European Brain Research Foundation. Americans, Karl Deisseroth, a 2005 Pioneer Awardee, and 2012 Transformative Research Awardee, and Edward S. Boyden, a 2007 New Innovator, and 2012 Transformative Research Awardee and four European scientists were awarded the 2013 prize for their contributions to the development of “optogenetics.” Optogenetics is a technique that merges the fields of optics and genetics using light to specifically control the activity of genetically selected neurons. This revolutionary technique has allowed scientists to begin to gain a better understanding of the way circuits of neurons carry out complex functions. This technique has broad application potential in research from studying fundamental activities, such as memory formation and breathing, to more complex disorders and diseases, such as addiction, psychiatric disorders, and Alzheimer’s disease.
Two NIH Common Fund High Risk-High Reward Awardees were named as winners of The Brain Prize for 2013, which is awarded by the Grete Lundbeck European Brain Research Foundation. Americans, Karl Deisseroth, a 2005 Pioneer Awardee, and 2012 Transformative Research Awardee, and Edward S. Boyden, a 2007 New Innovator, and 2012 Transformative Research Awardee and four European scientists were awarded the 2013 prize for their contributions to the development of “optogenetics.” Optogenetics is a technique that merges the fields of optics and genetics using light to specifically control the activity of genetically selected neurons. This revolutionary technique has allowed scientists to begin to gain a better understanding of the way circuits of neurons carry out complex functions. This technique has broad application potential in research from studying fundamental activities, such as memory formation and breathing, to more complex disorders and diseases, such as addiction, psychiatric disorders, and Alzheimer’s disease.
The Brain Prize, the world’s largest brain research prize, is awarded to scientists who have made an outstanding contribution to European brain research, and is awarded to scientists who have conducted research in Europe, or to scientists who have conducted research in collaboration with European scientists.
Read more about the Brain Prize and the 2013 Brain Prize press release.
Research enhances CLARITY of the brain in neurological disease
A major challenge to researchers studying the brain has been the inability to study a fully intact brain, from both the global and microscopic perspectives. For example, researchers might want to examine the way a chemical interacts with brain cells, or neurons, both in the context individual neurons and of long neural circuits across the entire brain. Previously, researchers had to choose either to study a fully intact brain, which involves a lengthy sample preparation process and potential structural dissimilarities compared to the original organ, or to study small sections of the brain where one can examine fine molecular and cellular interactions without knowledge of the original complex structure of the neural circuit and structure of the intact brain. Dr. Karl Deisseroth, a 2012 Transformative Research Awardee from Stanford University and his colleagues, have combined tools from the fields of chemical engineering, computational optics, and molecular genetics to develop a new technology to solve this problem by transforming a fully intact mouse brain into a completely clear gel-based brain with all structural and molecular integrity intact. This new technology, named Clear Lipid-exchanged Anatomically Rigid Imaging/immunostaining-compatible Tissue hYdrogel (CLARITY) replaces lipids, or fats, which make the brain appear white outside of the body, and help form the structural basis for the brain, with a clear gel-like material that binds to cells, proteins, DNA, RNA, and other small molecules in the brain. The result is a completely clear brain with a gel-like consistency that maintains the same molecular and cellular relationships and structure as the original brain.
The new process, allows researchers to fluorescently tag small molecules and specific cell types to visualize individual molecular interactions as well as entire cellular networks in 3-D providing insight at both the molecular and whole system level which previously was not possible. CLARITY can be used by researchers to observe fine details of brains from animals and human patients with symptomatic neurological diseases without losing the larger-scale whole system perspective, potentially leading to new insights into diseases that affect the brain.
Reference:
Chung K, Wallace J, Kim S-Y, Kalyanasundaram S, Andalman AS, Davidson TJ, Mirzabekov JJ, Zalocusky KA, Mattis J, Denisin AK, Pak S, Bernstein H, Ramakrishnan C, Grosenick L, Gradinaru V, Deisseroth K. Structural and molecular interrogation of intact biological systems. Nature, April 10, 2012. DOI: 10.1038/Nature12107.
Novel stem cell technique creates neurons
Drs. Kang Zhang and Sheng Ding, funded in part by the NIH Director’s Transformative R01 (T-R01) Award program, have unlocked the key to transforming human embryonic stem cells (hESCs) into a type of precursor cell that can be produced in large quantities and has the potential to become many different types of brain cells. Their findings, published in the May 17, 2011 issue of Proceedings of the National Academy of Sciences, represent a huge leap forward in stem cell science. hESCs, with their ability to become any cell type in the human body, hold great potential for repairing or replacing damaged tissues. However, a number of obstacles have prevented hESCs from fulfilling this promise. Scientists have faced challenges finding the right method to change hESCs into more specialized precursor cells, which can self-renew to produce large quantities of cells while also retaining the ability to become many different cell types within a specific tissue. Additionally, hESCs can cause the formation of tumors, which prohibits their use for therapeutic purposes. Drs. Zhang, Ding, and colleagues used a novel combination of small molecules to induce hESCs to become primitive neuronal stem cells (pNSCs), a cell type that can be directed to make many different types of neurons, or brain cells. Unlike hESCs, pNSCs did not induce tumor formation when injected into mice, which opens the door for potential therapeutic use. The researchers coaxed the pNSCs to form the types of neurons damaged by Parkinson’s disease and Lou Gehrig’s disease (amyotrophic lateral sclerosis; ALS), and suggest that pNSCs could be used to make many other types of neurons as well. This same method could be modified to direct hESCs to make other types of stem cells that could then be used to make heart, pancreas, or other tissue types. Future studies will need to examine how these cells could be used to treat a variety of human diseases.
Reference:
Li W, Sun W, Zhang Y, Wei W, Ambasudhan R, Xia P, Talantova M, Lin T, Kim J, Wang X, Kim WR, Lipton SA, Zhag K, and Ding S. Rapid induction and long-term self-renewal of primitive neural precursors from human embryonic stem cells by small molecules inhibitors. Proceedings of the National Academy of Sciences, 2011 May 17; 108(20): 8299-8304. PMID: 21525408.
Dual NIH Honors: Two Researchers Receive Pioneer and T-R01 Awards


Dr. Miguel A. Nicolelis at Duke University and Dr. J. Keith Joung at Massachusetts General Hospital and Harvard Medical School received “duals honors” through the Common Fund’ High-Risk Research program in FY 2010: an NIH Director’s Transformative TR01 award and Pioneer Award. Dr. Nicolelis is using his T-R01 Award to study dorsal spinal column stimulation as a novel alternative treatment of Parkinson's disease. Dr. Joung is identifying and applying new technologies that use molecular regulators to regenerate specific components of the nervous system and treat neurodegenerative diseases with his TR01 award.
- Four new DNA letters double life’s alphabet
Nature- DNA Gets a New—and Bigger—Genetic Alphabet
The New York Times - Seeing the Brain's Electrical Activity
EurekAlert! - In Vivo Label-Free Photoacoustic Flow Cytography and On-the-Spot Laser Killing of Single Circulating Melanoma Cells. He Y et al. Sci Rep. 2016 Dec 21;6:39616.
- Nanoscale Imaging of RNA with Expansion Microscopy. Chen F et al. Nat Methods. 2016 Aug 13;13(8):679-84.
- In the News: Nanoscale Microscopy Technique Allows Scientists to Pinpoint RNA Molecules in the Brain
- Optical Imaging of Individual Biomolecules in Densely Packed Clusters. Dai M, Jungmann R, Yin P. Nat Nanotechnol. 2016 Jul 4. doi: 10.1038/nnano.2016.95.
- In the News: From Super to Ultra-Resolution Microscopy
- Hypoxia as a Therapy for Mitochondrial Disease. Jain IH et al. Science. 2016 Apr 1;352(6281):54-61.
- Complementation of Mitochondrial Electron Transport Chain by Manipulation of the NAD+/NADH Ratio. Titov DV et al. Science. 2016 Apr 8;352(6282):231-5.
- Quantitative Super-Resolution Imaging with qPAINT. Jungmann R et al. Nat Methods. 2016 May;13(5):439-42.
- In Vivo Genome Editing Improves Muscle Function in a Mouse Model of Duchenne Muscular Dystrophy. Nelson CE et al. Science. 2016 Jan 22;351(6271):403-7.
- In Vivo Gene Editing in Dystrophic Mouse Muscle and Muscle Stem Cells. Tabebordbar M et al. Science. 2016 Jan 22;351(6271):407-11.
- In the News: Gene Editing Offers Hope for Treating Duchenne Muscular Dystrophy, Studies Find
- In the News: CRISPR Helps Heal Mice with Muscular Dystrophy
- Trace Incorporation of Heavy Water Reveals Slow and Heterogeneous Pathogen Growth Rates in Cystic Fibrosis Sputum.. Kopf SH et al. Proc Natl Acad Sci USA. 2016 Jan 12;113(2):E110-6.
- Extensively Drug-Resistant Tuberculosis in a Young Child after Travel to India. Salazar-Austin N et al. Lancet Infect Dis. 2015 Dec; 15(12):1485-91.
- Coupling of mRNA Structure Rearrangement to Ribosome Movement during Bypassing of Non-coding Regions. Chen J et al. Cell. 2015 November 19; 16(5):1267-80.
- Age-Dependent Electroencephalogram (EEG) Patterns During Sevoflurane General Anesthesia in Infants. Cornelissen L, Kim S-E, Purdon PL, Brown EN, and Berde CB. eLife. 2015 June 23; 4.e06513.
- The Ageing Brain: Age-Dependent Changes in the Electroencephalogram During Propofol and Sevoflurane General Anaesthesia. Purdon PL et al. Br J Anaesth. 2015 July; 115(Suppl 1):i46-i57.
- Ageing Delays Emergence from General Anaesthesia in Rats by Increasing Anaesthetic Sensitivity in the Brain. Chemali JJ et al. Br J Anaesth. 2015 July; 115(Suppl 1):i58-i65.
- Age-Dependency of Sevoflurane-Induced Electroencephalogram Dynamics in Children. Akeju O et al. Br J Anaesth. 2015 July; 115(Suppl 1):i66-i76.
- Groundbreaking Northeastern research sweeps the globe
- Promising antibiotic discovered in microbial ‘dark matter'
- Read more about this exciting new technology and watch videos of mouse brains transformed using CLARITY in the NIH Press Release
- Read about this exciting new technology in the NIH Director’s Blog
- Read the New York Times Article
- DNA Gets a New—and Bigger—Genetic Alphabet



