2011 Awardees

Adela Ben-Yakar, Ph.D. and Jonathan T. Pierce-Shimomura, Ph.D.
University of Texas at Austin
Project Title: High-Speed Opto-Fluidics to Screen Entire Nervous System in Aging and Disease
Grant ID: R01-AG-041135
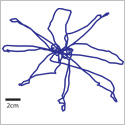
Figure describes a device that can manipulate small model nematodes (worms) in fluidic micro-channels. Populations of worms are treated with different drug compounds and then monitored for health of their entire nervous system at ultra-high speeds. This device will enable the discovery of new drugs to delay or prevent the progression of neurodegenerative diseases and aging.
Kwabena Boahen, Ph.D.
Stanford University
Project Title: Fully Implantable and Programmable Spike-Based Codecs for Neuroprosthetics
Grant ID: R01-NS-076460
Image shows the continuous cursor traces controlled by a brain-computer interface in a “center-out-and-back task” using eight targets arranged in a circle. Neural activity is recorded from 192 channels distributed across two microelectrode arrays implanted in the premotor and primary cortex. Data are decoded in real time to control the cursor.
Richard Cerione, Ph.D.; Hening Lin, Ph.D.; Robert Weiss, Ph.D.
Cornell University
Project Title: Succinylation and Malonylation as Novel Protein Modifications in Cancer
Grant ID: R01-CA-163255
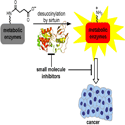
Image shows a sirtuin enzyme catalyzing the removal of a protein modification (lysine succinylation) from metabolic enzymes. This activity is required for the malignant growth of cancer cells. Small molecule inhibitors of the desuccinylation process may be a novel way to treat cancer.

Fred Gage, Ph.D.
The Salk Institute
Project Title: Single-Cell Approaches to Reveal How Jumping Genes Individualize Neural Circuits
Grant ID: R01-MH-095741
Identical twins are pictured in the foreground, while multi-colored neurons are depicted in the background, representing how mobile genetic elements can create unique mosaics of genomes within an individual’s brain.

Thomas Hartung, M.D., Ph.D.
Johns Hopkins University Bloomberg School of Public Health
Project Title: Mapping the Human Toxome by Systems Toxicology
Grant ID: R01-ES-020750
Outside-of-the-box toxicity testing for chemicals without the use of animals, represented by a street sign reading “Changes Ahead” below the word “Toxicology.”
Richard Honkanen, Ph.D.
University of South Alabama School of Medicine
Project Title: Methods to Enable Cholesterol Catabolism in Human Monocyte Derived Macrophages
Grant ID: R01-HL-110937
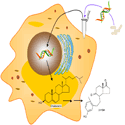
Image shows a schematic of genes being modified from bacteria, and then inserted into human immune cells. These genes give the immune cells the ability to “eat” cholesterol, suggesting a potential new treatment for cardiovascular disease.
![]()
Alan Jasanoff, Ph.D.
Massachusetts Institute of Technology
Project Title: Noninvasive Imaging-Based Electrophysiology Using Microelectronic Devices
Grant ID: R01-NS-076462
Image shows microscopic circuits that will enable monitoring of the brain's electrical activity using magnetic imaging techniques such as magnetic resonance imaging (MRI). Following injection into brains, the microdevices (shown in green) will convert neuronal voltage signals into tiny magnetic fields (shown in dashed blue lines) that will be detectable by MRI.
Thomas Kupper, M.D. and Rachael Clark, M.D., Ph.D.
Brigham and Women's Hospital / Dana Farber Brigham and Women's Cancer Center / Harvard Medical School
Project Title: Vaccination to Generate Protective Tissue Resident T Cells
Grant ID: R01-AI-097128
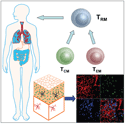
Vaccination through skin generates long-lived memory T cells, included central memory (TCM) and effector memory (TEM) T cells. Some of these memory T cells migrate into peripheral tissues, including skin, lung, and gastrointestinal tract, and become resident long-lived memory T cells (TRM) that protect against infection.

James Leckman, M.D. and Bruce Wexler, M.D.
Yale University
Project Title: Integrated Brain, Body, and Social Intervention for ADHD
Grant ID: R01-HD-070821
Figure illustrates the key components of an innovative and non-pharmacologic treatment for Attention Deficit Hyperactivity Disorder (ADHD) that combines cognitive and physical exercises. Part of the figure shows a child sitting at a classroom computer and doing one of the cognitive exercises. A second part of the figure shows a group of children engaged together in a group physical exercise in the gym that requires ball skills, concentration and memory. Last part of the figure shows a brain. Arrows back and forth between these images illustrate the “cross-training” of the brain that is the basis of the treatment.

Jeff Lichtman, M.D., Ph.D.; Markus Meister, Ph.D.; Joshua Sanes, Ph.D.; Sebastian Seung, Ph.D.
Harvard University and Massachusetts Institute of Technology
Project Title: High Resolution Connectomics of Mammalian Neural Circuits
Grant ID: R01-NS-076467
Color-coded reconstruction of three pyramidal neurons in the mouse brain. There are a number of axons making synapses on the beige neuron. Synapses are the sites of communication between neurons.
Todd McDevitt, Ph.D.
Georgia Institute of Technology / Emory University
Project Title: Stem Cell Morphogen Delivery Via Engineered Biomaterials
Grant ID: R01-AR-062006
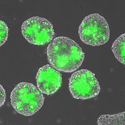
Small, round particles, on the order of 10 microns, can be mixed with stem cells of the same approximate size to form aggregates of the cells and materials. As stem cells secrete growth factors, the entrapped particles can capture the secreted molecules. The aggregates can then be disrupted in order to retrieve the particles, now loaded with the stem cell-derived factors. The stem cell factor-laden particles can then be injected at different sites of disease or injury in adult mammals in order to promote tissue repair and regeneration.

Vamsi Mootha, M.D.
Massachusetts General Hospital / Harvard Medical School
Project Title: Molecular Prostheses for Mitochondrial Disorders
Grant ID: R01-GM-099683
Image displays a high-resolution, high magnification electron micrograph of mitochondria isolated from heart.
Shuming Nie, Ph.D. and Sunil Singhal, M.D.
Emory University / Georgia Institute of Technology and University of Pennsylvania School of Medicine
Project Title: Contrast-Enhanced and Image-Guided Surgery of Lung Cancer
Grant ID: R01-CA-163256
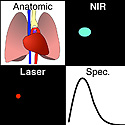
For high sensitivity and specificity in tumor detection, this system is equipped with 4 input channels, shown in 4 panels: (i) a high-definition video channel for anatomical color imaging; (ii) a laser light channel for real-time tracking of the laser beam position; (iii) a near-infrared (NIR) channel for wide-field imaging of wavelength-shifted contrast signals; and (iv) a spectroscopic channel for recording wavelength-resolved spectra of the contrast agents.
Jan Nolta, Ph.D.
University of California, Davis
Project Title: Direct Cell to Cell Transfer of MicroRNA for Tissue Repair
Grant ID: R01-GM-099688
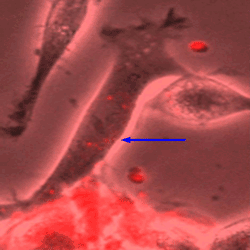
Image shows a mesenchymal stem cell (MSC) on the bottom of the figure transferring red-dye labeled pieces of RNA into a target neuronal cell (blue arrow).
F. Nina Papavasiliou, Ph.D.
Rockefeller University
Project Title: Building Novel Vaccines on a Borrowed Coat
Grant ID: R01-AI-097127
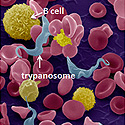
African trypanosomes are blood-borne parasites that use their surface coat as a decoy to trick the immune system into making long-lasting antibody responses. Image shows trypanosomes surrounded by immune cells (B cells). Trypanosome coats will be modified (shown in the middle panel) to trick the immune system into making therapeutic antibodies against disease-associated targets (inserted epitope, shown in the right panel).

Joseph D. Puglisi, Ph.D.
Stanford University School of Medicine
Project Title: Single Molecule Translational Profiling
Grant ID: R01-GM-099687

Margaret Elizabeth Ross, M.D., Ph.D. and Christopher E. Mason, Ph.D.
Weill Medical College of Cornell University
Project Title: Epigenome Interactions in Complex Neurogenetic Disorders
Grant ID: R01-NS-076465
Spina bifida is shown on the left, in magnetic resonance imaging (MRI), showing the open spine and exposed spinal nerves (arrow). This and other Neural tube defects (NTDs) are due to a complex interaction of genetic predisposition and environmental factors, including the addition of methyl groups to DNA. A major source of methyl groups is the vitamin, folic acid. On the right is a map of DNA methylation across all genomic DNA from one individual. Such maps from patients with spina bifida will be compared with healthy controls to determine which genes are under- or over- methylated in affected patients and could contribute to their risk of having an NTD.



