Science Highlights
Using Stem Cells to Catalyze Search for New Pain-Relieving Drugs
Chronic pain affects about 20% of US adults and is one of the top reasons for seeking healthcare. The condition, which is defined as pain that lasts at least 3 months, negatively affects quality of life and is the leading cause of disability in the US. Opioids are pain relievers that are commonly prescribed to treat pain. However, long-term use of opioids can lead to addiction, spurring the scientific community to research new drugs to treat pain.
which is defined as pain that lasts at least 3 months, negatively affects quality of life and is the leading cause of disability in the US. Opioids are pain relievers that are commonly prescribed to treat pain. However, long-term use of opioids can lead to addiction, spurring the scientific community to research new drugs to treat pain.
Scientists often rely on animal models to research pain treatments, but use of these models is not optimal because physiological differences between humans and animals can lead to drug candidates that do not work well or are toxic in humans. To facilitate the discovery of drugs that will be effective in humans, researchers supported by the Common Fund’s Regenerative Medicine Program made use of human pluripotent stem cells (hPSCs), which are cells that can become almost any type of human cell.
The goal of the Regenerative Medicine Program was to produce resources to accelerate stem cell-based therapies. Dr. Ilyas Singeç and colleagues developed a method (link is external) to generate nerve cells from hPSCs that initiate the sensation of pain. The nerve cells developed through this process have cellular features similar to nerve cells found in the human body and displayed biomarkers that are useful for drug development. The new method also allows for the production of large quantities of the nerve cells. This resource may accelerate research that aims to discover new treatments for chronic pain.
Reference:
- Scalable generation of sensory neurons from human pluripotent stem cells (link is external). T. Deng, V.M. Jovanovic, C.A. Tristan, C. Weber, P.-H. Chu, J. Inman, S. Ryu, Y. Jethmalani, J.F. de Sousa, P.Ormanoglu, P. Twumasi, C. Sen, J. Shim, S. Jayakar, H.-X. B. Zhang, S. Jo, W. Yu, T.C. Voss, A. Simeonov, B.P. Bean, C.J. Woolf, I. Singeç. Stem Cell Reports. 2023 Apr 1; 18:1.
CEPT: A recipe for a small-molecule cocktail that enhances the survival of stem cells
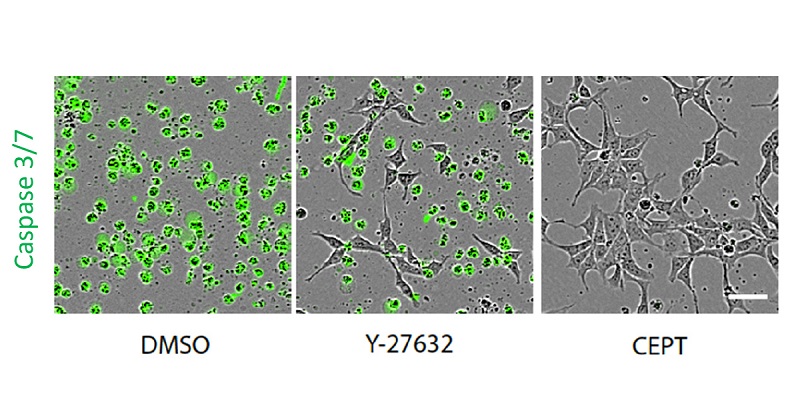
The combination of Chroman 1, Emricasan, Polyamines, Trans-ISRIB (CEPT), is a new resource that can help bring induced pluripotent stem cell technology closer to clinical applications.
When you hear “small cocktail,” stem cell research may be the furthest thing from your mind. In stem cell research, especially induced pluripotent stem cells (iPSCs) technology, small-molecule cocktails can be critical to improve cell survival. iPSCs are a special type of stem cell derived from skin or blood cells that have been reprogrammed into a stem cell-like state where they can become any cell type in the body. iPSCs-based therapy is already being tested in clinical trials to treat retinal degeneration, heart failure, and neurological diseases [1] and have the potential to treat chronic wounds [2]. However, the development of iPSC-based therapy can be difficult, partly due to cell-culture induced stress that affects the quality of cells. The lack of high quality iPSCs can limit production of the large amounts of cells needed for disease research, tissue engineering, drug development and regenerative medicine. To address technical challenges that impede the use of iPSC technology for clinical applications, NIH launched the Stem Cell Translation Laboratory (SCTL), part of the NIH Common Fund's Regenerative Medicine Program and housed within the National Center for Advancing Translational Sciences (NCATS).
A team of researchers at SCTL, led by Dr. Ilyas Singeҫ, tested more than 15,000 U.S. Food and Drug Administration-approved drugs and compounds that led to identification of small-molecule cocktails that would protect iPSCs from cellular stress and improve cell survival. They found CEPT, a four-factor drug combination – or cocktail – made up of chroman 1, emricasan, polyamines, and trans-ISRIB that preserves cell survival through processes including protection from DNA damage and lowering of oxidative stress. Additionally, the team demonstrated that treating iPSCs with CEPT makes establishing new types of iPSCs more feasible and producing larger quantities possible. Thus, the CEPT cocktail has the potential to support up-scaling of iPSCs for clinical use.
Findings from this work are presented in Nature Methods [3] and indicate that CEPT is a safeguard for iPSCs. Though additional research is needed, using the small molecule cocktail of CEPT treatment may become a standard method to efficiently generate iPSCs and move the field of stem-cell research forward. This new approach is a building block to develop advanced strategies for drug discovery and accelerate breakthroughs in the development of stem cell-based therapies for complex diseases.
Read more about this work in new articles.
References:
- Deinsberger J, Reisinger D, Weber B. Global trends in clinical trials involving pluripotent stem cells: a systematic multi-database analysis. NPJ Regen Med. 2020 Sep 11; 5:15.
- Gorecka J, Kostiuk V, Fereydooni A, Gonzalez L, Luo J, Dash B, Isaji T, Ono S, Liu S, Lee SR, Xu J, Liu J, Taniguchi R, Yastula B, Hsia HC, Qyang Y, Dardik A. The potential and limitations of induced pluripotent stem cells to achieve wound healing. Stem Cell Res Ther. 2019 Mar 12;10(1):87.
- Chen Y, Tristan CA, Chen L, Jovanovic VM, Malley C, Chu PH, Ryu S, Deng T, Ormanoglu P, Tao D, Fang Y, Slamecka J, Hong H, LeClair CA, Michael S, Austin CP, Simeonov A, Singeç I. A versatile polypharmacology platform promotes cytoprotection and viability of human pluripotent and differentiated cells. Nat Methods. 2021 May 3.
Stem Cell-Based Treatment Used to Prevent Blindness in Animal Models of Retinal Degeneration
Age-related macular degeneration (AMD) is the most common cause of blindness among the elderly. With limited treatment options for AMD patients, scientists turned to regenerative medicine approaches, pursuing an induced pluripotent stem cell (iPSC) based therapy for AMD. IPSCs are cells taken from patient tissues and coaxed into a stem cell-like state where they can be become any cell type in the body. In a study led by Dr. Kapil Bharti, a Regenerative Medicine Program-funded investigator, dying retinal pigment epithelial (RPE) cells in pigs and rats with retinal deterioration were replaced with human iPSC-derived RPE cells. The replacement cells helped nurture photoreceptors, the light-sensing cells in the retina. Both healthy RPE and photoreceptor cells are necessary to maintain vision. With both cell types now in place, the investigators observed that the iPSC-based therapy successfully prevented blindness in the animals. The findings from this study will inform a first-in-human clinical trial to treat AMD.
- Watch Dr. Bharti explain his research: https://youtu.be/k6DTj597Gv8.
Reference:
- Clinical-grade stem cell–derived retinal pigment epithelium patch rescues retinal degeneration in rodents and pigs. Sharma R, Khristov V, Rising A, Jha BS, Dejene R, Hotaling N, Li Y, Stoddard J, Stankewicz C, Wan Q, Zhang C, Campos MM, Miyagishima KJ, McGaughey D, Vilasmil R, Mattapallil M, Stanzel B, Qian H, Wong W, Chase L, Charles S, McGill T, Miller S, Maminishikis A, Amaral J, Bharti K. Science Translational Medicine. January 16, 2019 (Note: full text of the article may require institutional access)
In the News:
- NIH researchers rescue photoreceptors, prevent blindness in animal models of retinal degeneration-NEI Press Release
- Macular degeneration trial will be first human test of Nobel-winning stem cell technique (link is external)-STAT
- Moving Closer to a Stem Cell-Based Treatment for AMD- NIH Director's Blog
Kapil Bharti’s Therapeutic Challenge Award to develop a stem-cell based therapy for macular degeneration – a three-year update
Overview: Therapeutic Challenge Awardee Dr. Kapil Bharti of the National Eye Institute (NEI) and his research team have made major strides towards developing an inducible pluripotent stem cell (iPSC)-based therapy to treat age-related macular degeneration (AMD), a common cause of blindness. In AMD patients, Retinal Pigment Epithelium (RPE) cells – responsible for nourishing and supporting the retinal photoreceptor (light-sensitive) – become damaged and lose function. Replacement of damaged RPE cells with functional iPSC-derived RPE cells is a viable treatment believed to restore or improve vision. iPSCs are stem cells that have been reprogramed from adult cells and can develop into any cell type in the body. For clinical applications, iPSCs are to be manufactured using strict guidelines called current good manufacturing practice (cGMP) to ensure quality and clinical compatibility standards of cells. Dr. Bharti’s team has developed a streamlined manufacturing process to generate transplant-ready clinical-grade RPE cells from iPSCs. His team is currently working to complete the pre-clinical animal toxicity, efficacy, and transplantation device compatibility studies in preparation for submitting a phase I Investigational New Drug (IND) application with the U.S. Food and Drug Administration in late 2017 or early 2018. Learn more about Dr. Bharti’s research at NEI.
Project highlights: Over the past three years, Dr. Bharti’s team has overcome many challenges in bringing an iPSC-based therapy for AMD much closer to the clinic. Notable successes include (click link to expand and hide descriptive text):
Developing a biodegradable scaffold for growing, maturing and implanting RPE cell monolayers
Ensuring every step of the manufacturing process is cGMP-compliant

This is a scanning electron microscope image of an RPE monolayer growing on top of a biodegradable scaffold (blue, left side of image). The polarity of the RPE cells is clearly visible (brown, bottom of cells in middle of image; green, top of cells on right side of image).
NIH Center for Regenerative Medicine and NIH Clinical Center join forces in an effort to harmonize informed consent for iPS cell-based research and therapies
The NIH Center for Regenerative Medicine has collaborated with the NIH Clinical Center Department of Bioethics on a manuscript that considers the challenges to obtaining informed consent to derive induced pluripotent stem (iPS) cells from donated tissue samples for research and therapeutic purposes. The manuscript provides concrete recommendations, along with a model consent form, in an effort to broadly harmonize informed consent, which currently varies considerably between tissue collection sites.
The promise of iPS cells in clinical therapies to replace damaged or diseased tissues, and as a resource for understanding a wide range of diseases and discovering candidate therapeutic drugs, is huge. However, to maximize their utility while safeguarding the donors that provide the starting material (e.g. skin) that the iPS cells are derived from, it is critical that informed consent is carefully considered now while the field is still nascent. This is particularly challenging when all the potential downstream uses of iPS cells are not yet know and can result in narrowly restricting the informed consent to a specific application at hand. Conversely, re-contacting donors/study participants indefinitely to re-consent to additional uses of the iPS cells, or to collect additional samples or information on their health, is not ideal either. This manuscript makes a cogent argument for a middle ground that balances the perspectives of a variety of stakeholders and arrives at a model consent form for the prospective collection of fresh specimens from which to derive iPS cells. Some important features include:
- Research purposes are “open-ended” within the boundaries of all applicable laws and policies and may include transplanting cells or tissues made from iPS cells in to another patient to treat a disease
- The iPS cells and participant’s medical information will be shared with other researchers to benefit medical research and society
- Donors may be re-contacted for additional samples, medical information, or to consent to additional iPS cell uses not originally anticipated, but they do not need to oblige
- A provision is included for donors to opt out of being re-contacted by researchers for any reason
- Pediatric re-consent is incorporated to allow children to be informed upon reaching adulthood of the ability to review the previous consent and continue to participate in the study or not
- Explicit language is included indicating that direct medical benefits for the donor are unlikely and no financial benefits will result from any commercial products developed from the iPS cells
- Withdrawal at different stages of the research process is allowed. A request can be made to destroy any leftover original samples; however, any iPS cells already derived cannot be destroyed and any shared with other researchers cannot be retrieved, though the codes that link the sample to the donor can be removed
The implementation of a consistent approach to informed consent for iPS cell derivation and use across research/medical institutions in these early days as proposed in this manuscript could potentially be invaluable to providing stem cell researchers with crucial access to high-quality, thoroughly documented materials and resources.
Reference:
Lowenthal J, Lipnick S, Rao M, Hull SC. Specimen collection for induced pluripotent stem cell research: Harmonizing the approach to informed consent. Stem Cells Translational Medicine. Published online May 8, 2012; doi: 10.5966/sctm.2012-0029.
Current good manufacturing practices induced pluripotent stem cell line now available to enable development of new therapies and accelerate early-stage clinical research
Induced pluripotent stem cells (iPSCs) represent a powerful new avenue for potentially treating ailments ranging from Alzheimer's disease to spinal cord injury. Significant progress with stem cell therapy in mice is already underway; for instance, researchers have reversed diabetic conditions in mice using iPSC-generated insulin-producing beta-cells. Translating these studies into humans is the next challenge. The RMP supported, through a contract with Lonza Walkersville Inc., the development of a current good manufacturing practice (cGMP) clinical-grade iPSC line from human umbilical cord blood CD34+ cells. cGMP is a set of stringent regulations enforced by the US Food and Drug Administration that ensures each batch of cells produced will meet quality and safety standards required for potential clinical use. These cells were fully characterized for safety, sterility, plasmid clearance, endotoxin levels, karyotype and many other factors and are described in two publications by Behnam Baghbaderani and colleagues in Stem Cell Reports and Stem Cell Rev. The cGMP clinical-grade iPSC line – along with a parallel, identical, non-cGMP research grade iPSC line – is available to the research community through RUCDR Infinite Biologics (link is external) at Rutgers University.
The Common Fund Issues New FY 2012 Awards in Regenerative Medicine
Thirteen NIH Intramural Research Program (IRP) laboratories have been funded on a competitive basis in fiscal year 2011 to conduct pilot projects in support of the NIH CRM mission.
The Common Fund Issues New FY2011 Awards to Spur Regenerative Medicine Research
Thirteen NIH Intramural Research Program (IRP) laboratories have been funded on a competitive basis in fiscal year 2011 to conduct pilot projects in support of the NIH CRM mission.
The Common Fund Issues First Awards in FY2010
Eleven laboratories in the NIH Intramural Research Program (IRP) are being supported to conduct pilot studies on the clinical applications of induced pluripotent stem cells (iPSCs).



